Take a moment and consider something remarkable.
The oxygen you breathe, the calcium in your bones, and the iron flowing through your blood were not created on Earth. Long before our planet existed, these elements were forged in the hearts of ancient stars and scattered across space during violent stellar explosions.
Every atom in your body has traveled an extraordinary journey through the cosmos. In a very real sense, we are made from the recycled remains of stars.
But how did that happen?
To understand where the elements that built our planet and our bodies came from, we need to go back to the beginning of the universe.
The First Elements
Nearly 13.8 billion years ago, the universe began with the Big Bang. In the first few minutes after this event, the universe cooled enough for the simplest atomic nuclei to form. During this brief period only a small number of elements were created, primarily hydrogen and helium, along with trace amounts of lithium.
Hydrogen quickly became the dominant element in the cosmos, making up roughly 75% of ordinary matter. At this early stage, the universe contained none of the elements required for life. There was no carbon, oxygen, nitrogen, calcium, or phosphorus. Those elements would not appear until the first stars began to shine.
The Birth of Stars
Over time, gravity began gathering vast clouds of hydrogen gas. As these clouds collapsed, pressure and temperature increased at their centers until nuclear fusion ignited. The first stars were born.
Inside these stellar furnaces, hydrogen atoms fused together to form helium, releasing enormous amounts of energy. This process powered the light and heat of the earliest stars and began the gradual transformation of the universe.
As stars evolved, new fusion reactions began creating heavier elements. Helium fused into carbon. Carbon fused into oxygen. In the most massive stars, this process continued step-by-step through the periodic table until reaching iron.
Iron marks a critical turning point in stellar life.
Fusion reactions that create elements lighter than iron release energy, but creating elements heavier than iron requires energy rather than producing it. When a massive star builds an iron core, it can no longer support itself against gravity, and the star collapses under its own immense weight.
The Death of Stars
When the core of a massive star collapses, the outer layers rebound outward in a tremendous explosion known as a supernova. For a brief moment, the exploding star can shine brighter than an entire galaxy.
During this violent process, many of the heavier elements are created, including calcium, phosphorus, gold, silver, and uranium. The explosion blasts these newly formed elements outward into space, where they mix with surrounding clouds of gas and dust.
Each generation of stars enriches the universe with new material. Over time, the cosmos becomes chemically richer, gradually building the elements needed for planets, oceans, and eventually life.
How Did We End Up Here?
About 4.6 billion years ago, one of these enriched clouds of gas and dust collapsed to form our solar system. Astronomers believe a nearby supernova may have helped trigger this collapse by compressing the cloud and causing it to begin forming new stars.
Evidence for this event comes from radioactive isotopes found in meteorites—particularly iron-60, which can only be produced in supernova explosions. From this swirling nebula formed the Sun, the planets, asteroids, and comets. Over time, dust grains collided and merged to form larger bodies, eventually giving rise to the rocky worlds of the inner solar system, including Earth.
Our Elemental Design
The human body is composed primarily of just a handful of elements. In fact, six elements make up about 99% of the mass of the human body.
Oxygen accounts for roughly 65% of our mass, followed by carbon at about 18% and hydrogen at around 10%. Nitrogen contributes roughly 3%, while calcium and phosphorus make up smaller but essential portions of our bodies.
Each of these elements has a different cosmic origin:
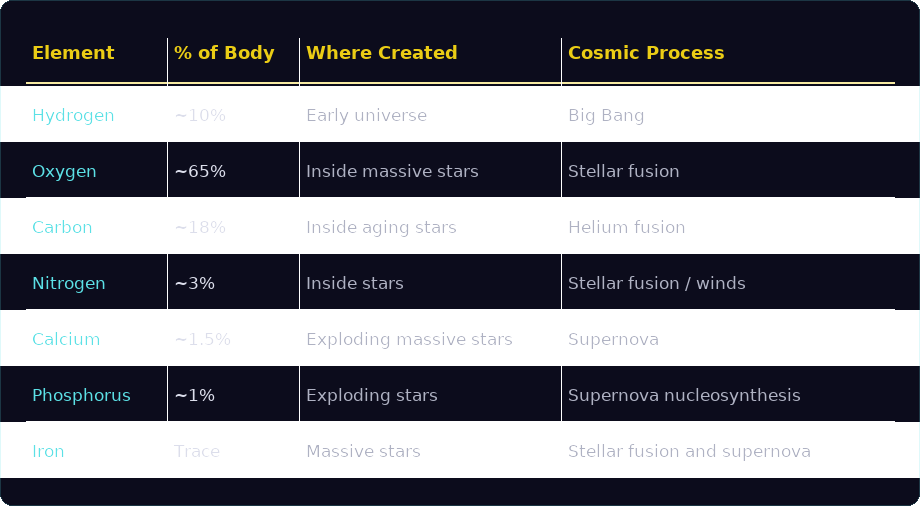
Together, these elements form our biological architecture. Oxygen and hydrogen combine to create water. Carbon forms the structural framework of DNA and proteins. Calcium strengthens our bones, while phosphorus plays a central role in cellular energy (ATP) and our genetic structure.
Atoms of Creation
The elements that make up our bodies have been recycled through multiple generations of stars. A single atom of oxygen or carbon may have existed inside several different stars before becoming part of our planet.
Over billions of years stellar births and stellar deaths seeded the universe with increasingly complex material. Eventually one small region of this cosmic cloud formed a star we now call the Sun. Around it a small rocky planet formed, and on that planet chemistry slowly evolved into biology.
Every atom in your body once traveled through the cosmos.
Some were born in the first minutes of the universe. Others were forged deep inside stars. Still others were created during the explosive deaths of those stars.
The atoms in your body may have once burned inside a distant star billions of years ago. They traveled through space, became part of our planet, and eventually became part of you.
Every person on Earth shares this same origin story.
Beneath all our differences, we are made from the same ancient material of the universe.
"We are made of star-stuff." - Carl Sagan

